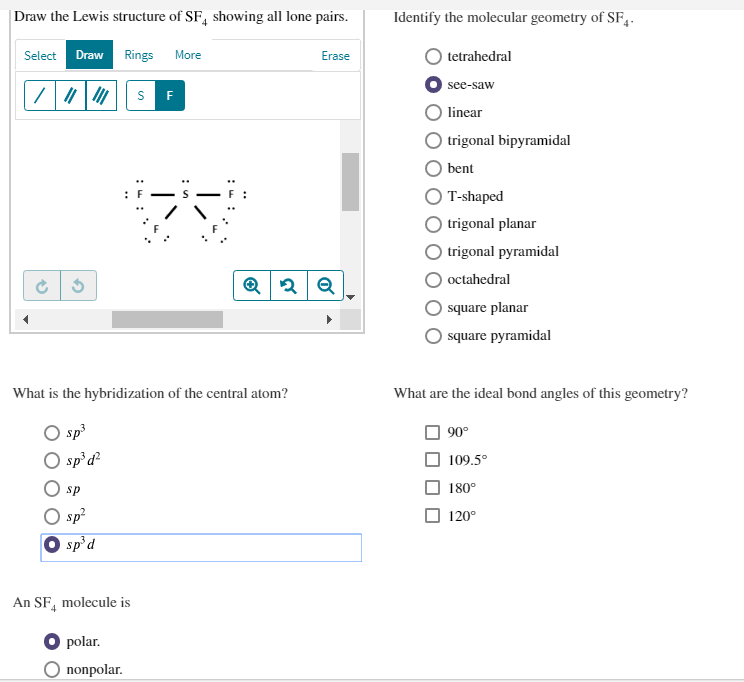
When the central atom is surrounded by bonded electron pairs of dissimilar atoms repulsive interactions are not equivalent and hence geometry is not regular.Examples of AX4E type molecules: SF4, TeCl4, SCl2F2, IF4+, IO2F2, etc. The central atom in AX 4 E-type molecules has sp 3 d hybridization. The molecular geometry or shape of AX 4 E-type molecules is seesaw. The presence of lone pair of electrons on the central atom causes some distortions in the expected regular shape of the molecules. The ideal electronic geometry of AX 4 E-type molecules is trigonal bipyramidal. The 'shared pairs' of electrons are also called bond pairs of electrons. Electron pairs around the molecule's central atom can be shared or can be lone pairs.In order to minimize the repulsive forces between them, electron pairs around the central atom, tend to stay as far away from each other as possible.The shape of the molecule is determined by the total number of electron pairs(bonding and nonbonding) around the central atom and the orientation of these electron pairs in the space around the central atom.JEE Main 2022 Question Paper Live Discussion.Difference Between Selling And Marketing.TS Grewal Solutions Class 11 Accountancy. 
Typically the bond distance to the axial ligands is longer than to the equatorial ligands. The equatorial pair of ligands is situated in a plane orthogonal to the axis of the axial pair. The axial pair lie along a common bond axis so that are related by a bond angle of 180°. TS Grewal Solutions Class 12 Accountancy Compounds with disphenoidal (see-saw) geometry have two types of ligands: axial and equatorial.CBSE Previous Year Question Papers Class 12.CBSE Previous Year Question Papers Class 10.NCERT Solutions For Class 6 Social Science Study with Quizlet and memorize flashcards containing terms like Give the molecular geometry and number of electron groups for SF4, Given the molecular geometry and number of electron groups for BrF5, How many of the following molecules are polar (PCL5, COS, XeO3, SeBr2) and more.NCERT Solutions for Class 7 Social Science.NCERT Solutions for Class 8 Social Science.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
